MASTER DAPT


Click here to download the summary of MASTER DAPT.

Get insight from interview about optimal DAPT duration with Dr. Muramatsu.
Overview
Investigator-initiated
global randomized study
global randomized study
4,579 patients,
140 hospital worldwide, 30 countries
140 hospital worldwide, 30 countries
All-comers trial in
HBR patients after PCI
HBR patients after PCI
Abbreviated vs prolonged
DAPT in patients treated with
the Utltimaster™and Ultimaster™ Tansei™ DES
DAPT in patients
treated with the
Ultimaster™ and
Ultimaster™Tansei™
DES
treated with the
Ultimaster™ and
Ultimaster™Tansei™
DES
Study Design
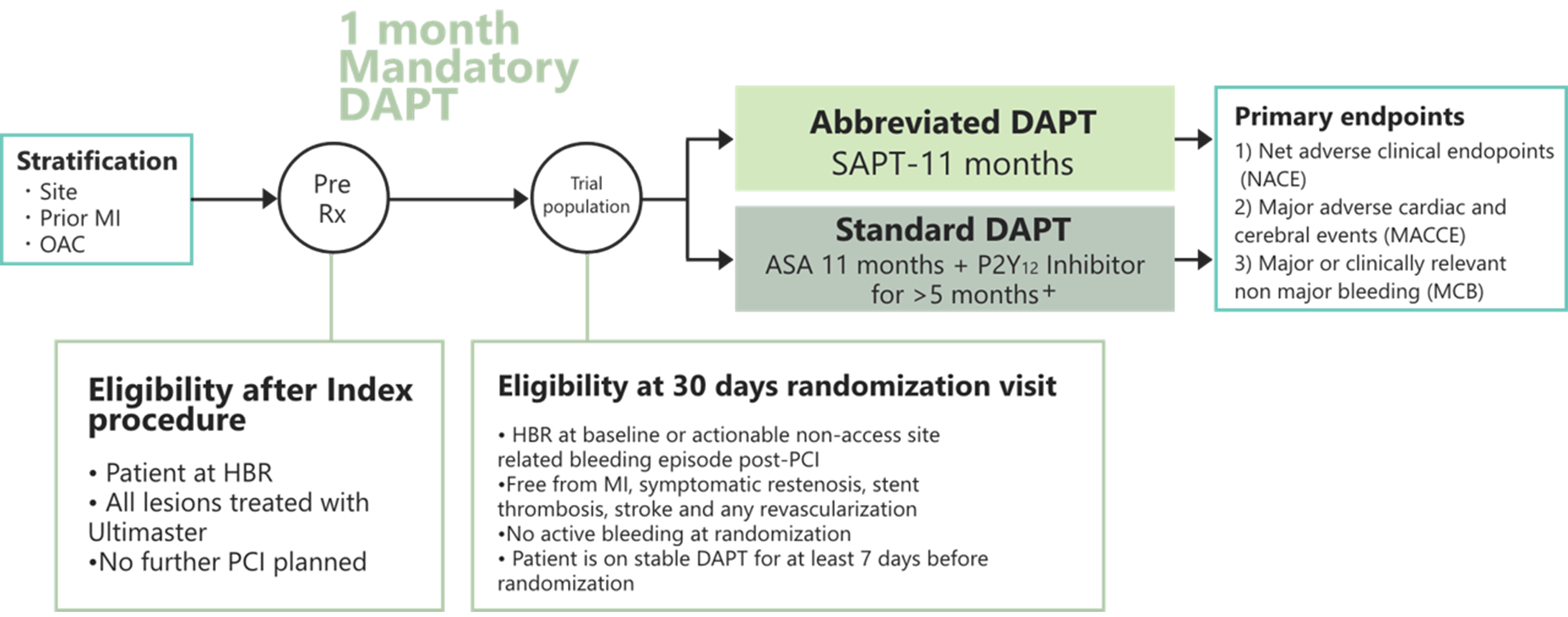
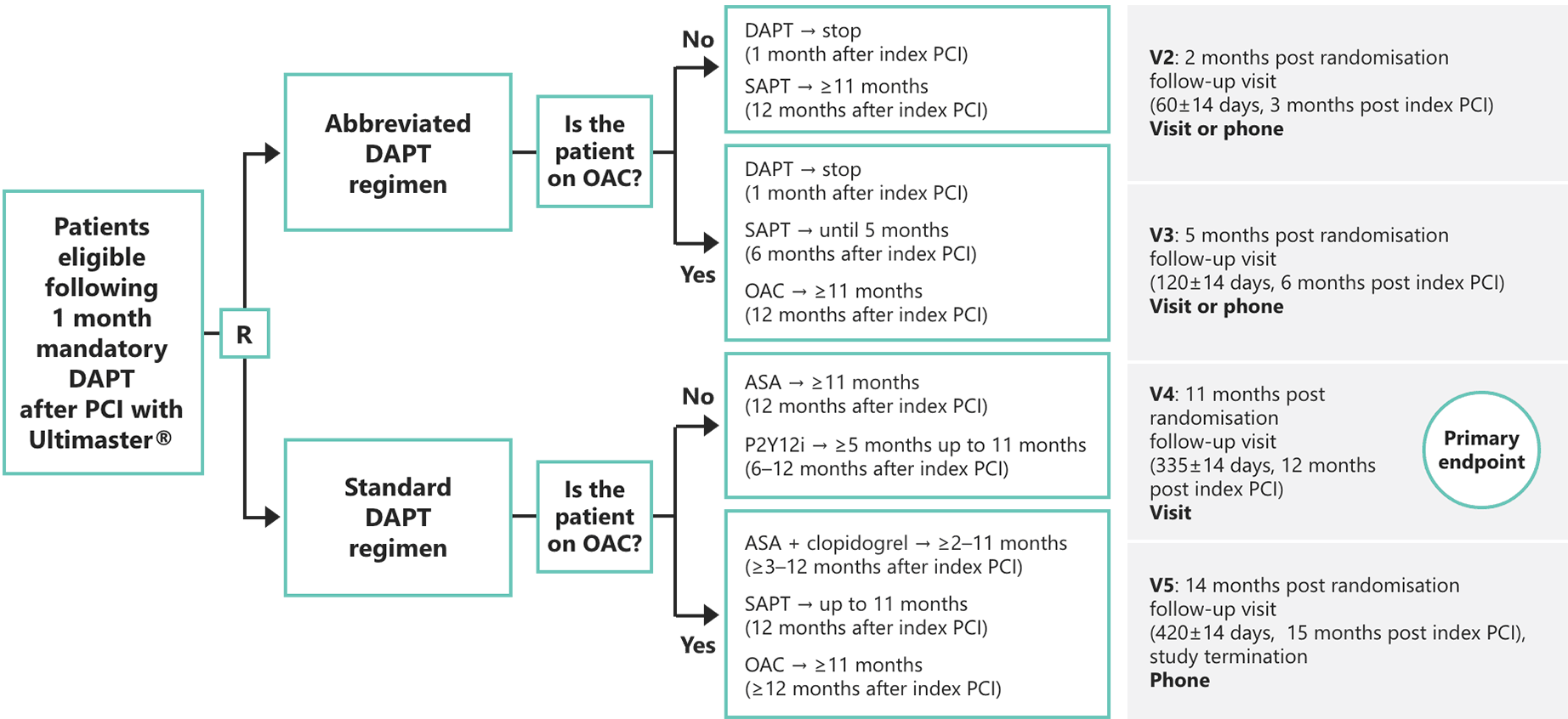
Primary Endpoint
・Net adverse clinical events (NACE) defined as a composite of death from any cause, myocardial infarction, stroke or major bleeding.
・Major adverse cardiac and cerebrovascular events (MACCE), defined as a composite of death from any cause, myocardial infarction, stroke or major bleeding.
・Major or clinically relevant non-major bleeding (MCB), defined as BARC 2, 3 or 5 bleeding.
Result
NACE
Difference in cumulative incidence,
0.23 percentage points (95% CI, 1.80 to 1.33)
P<0.001 for noninferiority
0.23 percentage points (95% CI, 1.80 to 1.33)
P<0.001 for noninferiority
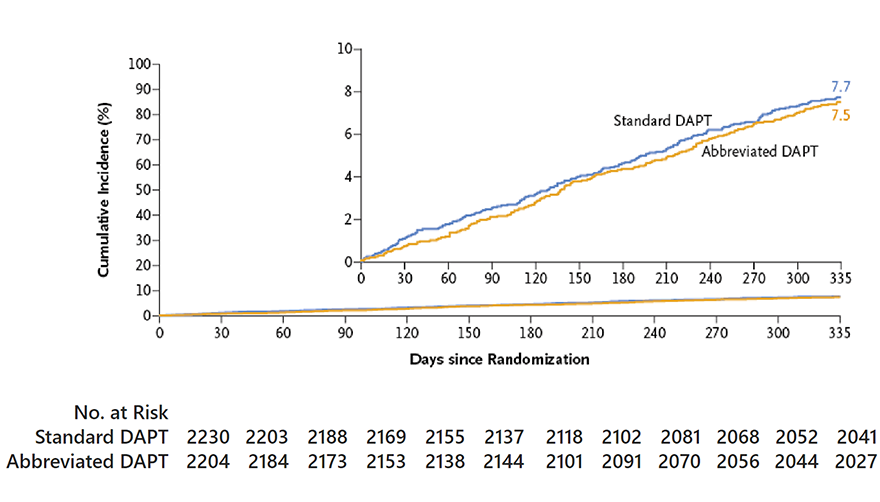
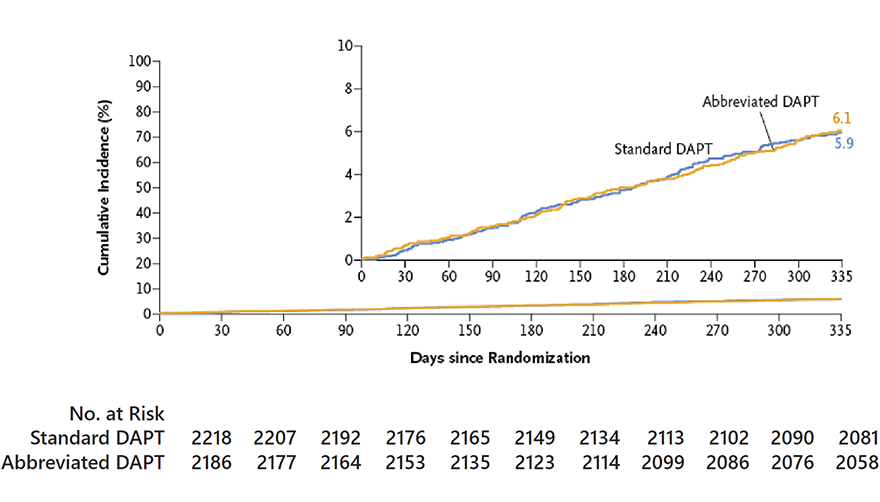
MACCE
Difference in cumulative incidence,
0.11 percentage points (95% CI, 1.29 to 1.51)
P=0.001 for noninferiority
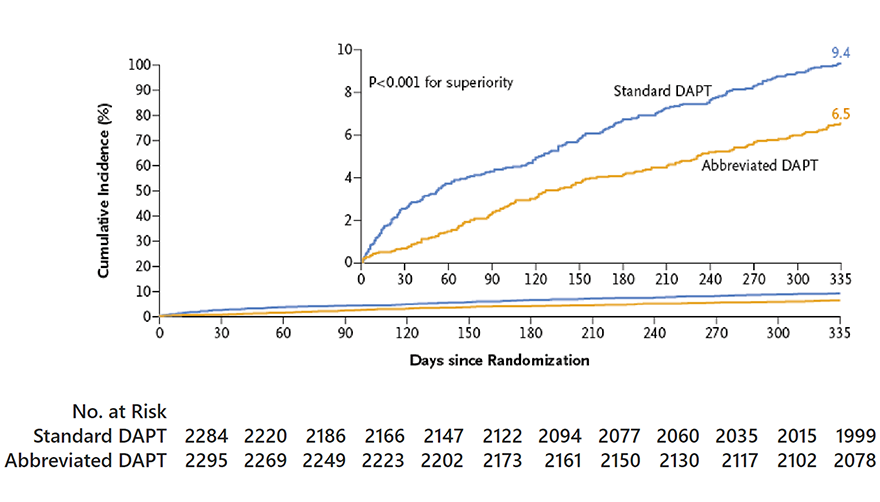
Major or clinically relevant non-major bleeding
Difference in cumulative incidence,
-2.82 percentage points (95% CI, 4.40 to 1.24)
P<0.001 for superiority
-2.82 percentage points (95% CI, 4.40 to 1.24)
P<0.001 for superiority
Special Topics
The Summary of MASTER DAPT
Dr.Marco at ESC Congress 2021
MASTER DAPT study is sponsored by the European Cardiovascular Research Institute (ECRI, Rotterdam, The Netherlands) and supported with a restricted research grant by Terumo Europe.