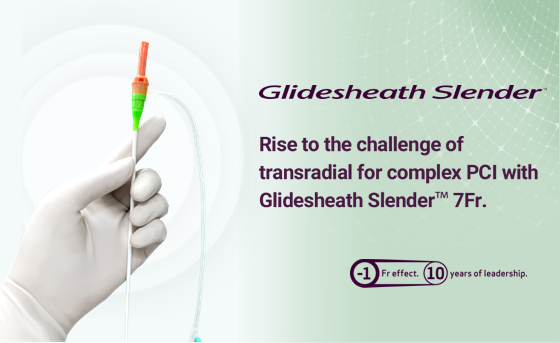
Terumo’s journey in interventional medicine began in 1985 with the launch of RADIFOCUS™ Guide Wire M. Now, over three decades later, our dedication to intervention, where access devices serve as a cornerstone, has not waned. Learn more from this conversation with our three integral members of Access team.

Tomomi Kitagawa
Senior Product manager, Access,
Global Marketing
Career began in R&D, engaged in drug-related work for DES. Later moved into marketing, before working in a wide range of areas including angiographic catheters, closure devices, and AI imaging. Currently working with radial vascular closure devices.

Kei Ozawa
Research Engineer, Ashitaka factory
After joining Terumo, gained experience as part of the life cycle management team for peripheral support catheters and guiding catheters. Currently involved in new product developments for sheaths and abdominal catheters.

Chihiro Mizuo
Research Engineer, Ashitaka factory
As part of the new product development team, participated in conceptual planning of guide wires designed for peripheral arteries, before transitioning to the team focused on angiographic catheters. Currently responsible for regulatory compliance, integral to ensuring our products remain on the market.
- How would you describe access devices?
Tomomi Kitagawa (TK): At Terumo, we’ve been providing access devices for almost forty years now, and gradually our products have gained a reputation for quality and reliability among healthcare professionals around the globe. So when it comes to developing access products, it’s not so much about revolutionizing them, but more about preserving the features that healthcare professionals have come to trust, while also finding ways to incorporate improvements. 
Kei Ozawa (KO): Exactly. Access devices are the unsung heroes of interventional treatment, quietly holding down the fort behind the scenes. They may not capture the spotlight, but their foundational role in medical treatment is undeniable.
Chihiro Mizuo (CM): Looking back on advancements in the 1990s and early 2000s, you get a real sense of the historical evolution and legacy of our access devices. Our broad product lineup is a testament to our constant adaptability, showing how we have continued to keep pace with and adapt to the changing needs of doctors.
- What challenges are typical when dealing with products that have such a rich history?

Take data collection, for example. When regulations change, sometimes previous methods are made non-compliant, or new criteria gets introduced to the mix. Coupled with the individual regulations of every country, with their different requirements for each package, ensuring global compliance becomes its own daily adventure.
But despite this ever-changing environment, our dedication stands firm. After all, the more stringent the regulations, the better the quality, and meeting those standards means we’re delivering the best possible product to our customer.
TK: One of the issues with long-selling products is that their inherent benefits and features can fade into the background. Getting these benefits across to customers can be its own challenge. For example, our universal shape catheter can lead to reduced procedural time1, less radiation exposure2, and lower costs3. Making sure these benefits are communicated effectively is another way for us to contribute to clinical settings.
- What kind of challenges come into play when developing a new product?
As costs of treatment increase, our products need to adapt. I also see the demand for sustainable products developing in the future, and a need to consider more environmentally-friendly methods and materials. Ultimately, my objective is to work within these confines to develop something that provides even more value.

And it’s not just about observing clinical settings—visiting the production line is just as important. Most issues aren’t going to be resolved from behind a desk. Associates involved in production have a unique understanding and perspective that’s distinct from mine, and engaging with them always provides a fresh viewpoint. It means problems can be tackled more swiftly, and we can be more proactive in finding a solution. The associates working in production don’t mince words—they tell it like it is and that’s exactly what’s needed!


I remember this one time, a co-worker had made a mistake and I was on hand to help troubleshoot. It was a Friday and even though the workday was technically over, I was still working on the issue. So, I was surprised when my co-worker clocked out as soon as the standard workday ended. Where I thought that wrapping it up as early as possible was the right approach, he was content to shelve it until next week.
In another instance, a co-worker mentioned that my Japanese manager sent mixed signals, and it was impossible to know what he was thinking. At first, I wasn’t sure how to respond. Later, however, I realized that my co-worker was used to a workplace culture characterized by open and candid sharing of opinions. In contrast, my manager communicated in a way more typical to Japan, one where his intentions were cushioned. I could see how this could be confusing for my co-worker, and it was a reminder that I could be more honest and frank when sharing my own doubts and opinions.
These experiences have shaped my understanding, teaching me to recognize and accept the different mindsets among individuals. I believe that embracing diversity is a step toward genuine mutual respect. When I feel respected and valued, I’m more inclined to engage and collaborate. And that, in my view, is vital for working effectively together.
- Which of the three personalities, Imaginative Ideas, Attention to Details, Never Give Up, resonates with you as an individual?
Imaginative Ideas

Attention to Details

Never Give Up, Imaginative Ideas

*Information accurate as of October 2023.
Other stories

Less-invasive solution for interventional oncology - learn how we push the boundaries in introducing RADIFOCUS™ Glidecath™, the first Terumo device for radial access to visceral.

Passion across three generations - engineers of Ultimaster™ talk about how we push the boundaries in developing each new generation of DES.