e-Ultimaster
Overview
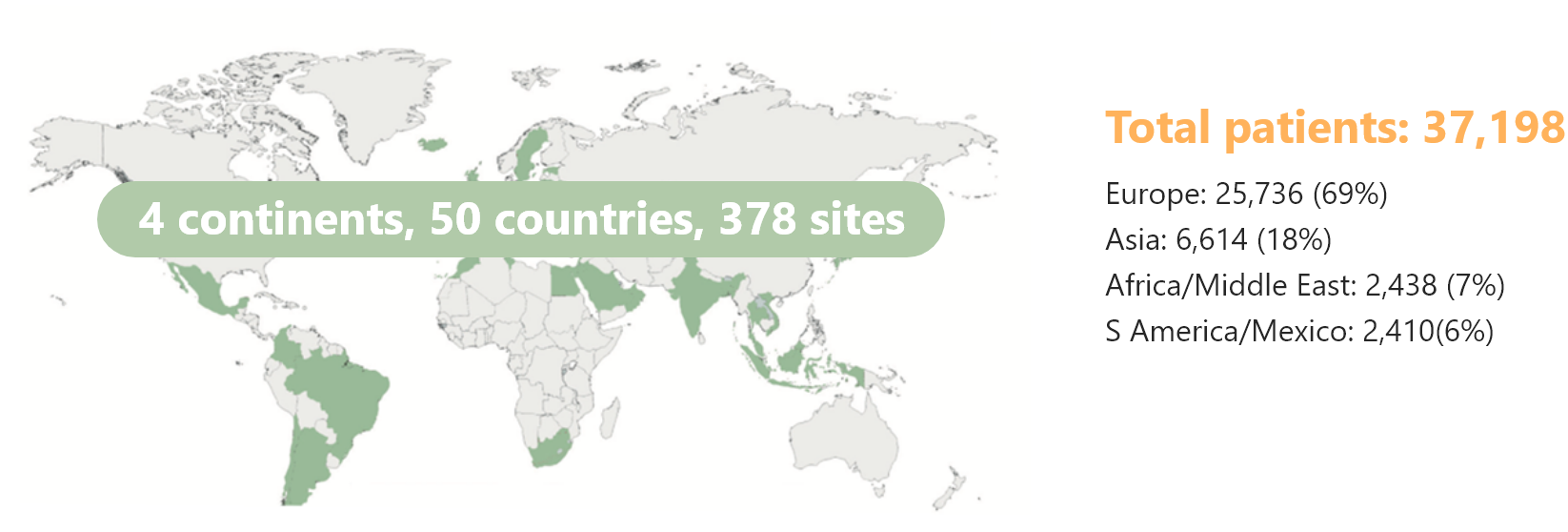
- 37,198 patients enrolled: The biggest real world BP-DES registry in the world.
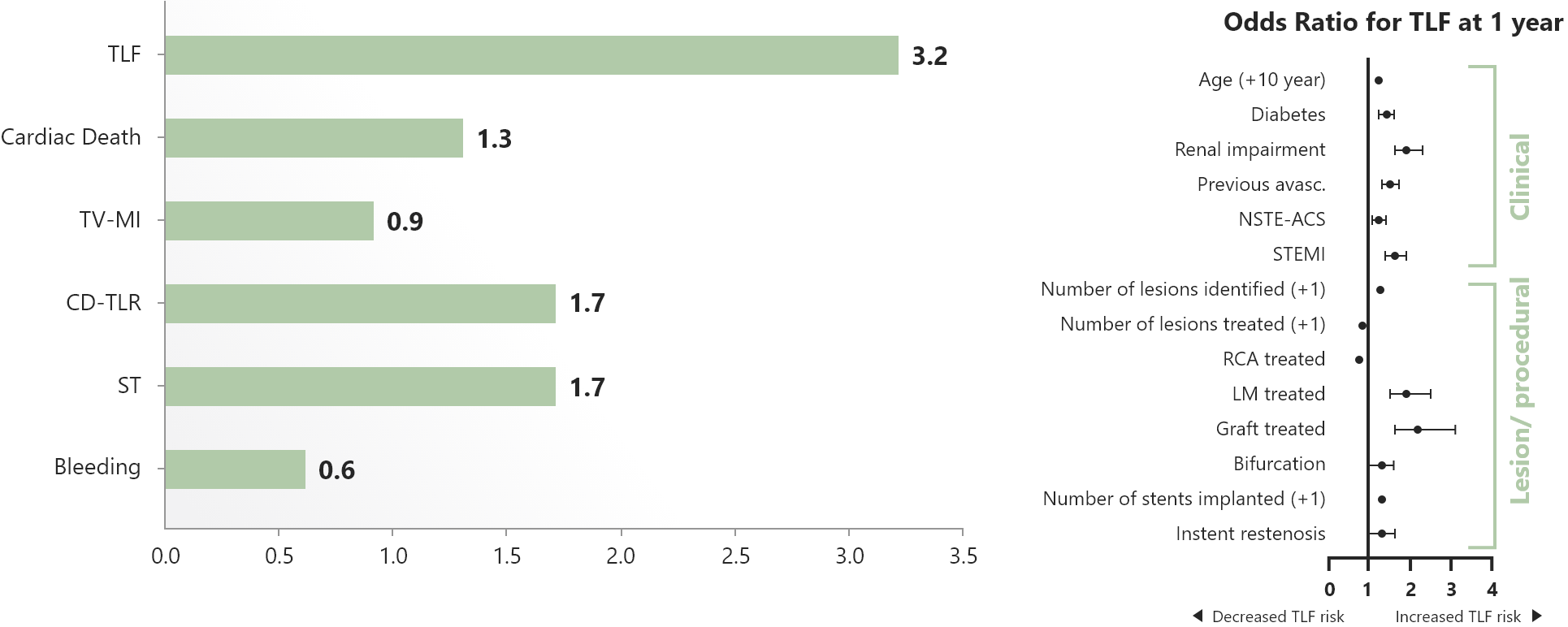
- Excellent performance with a TLF rate < 5% in all continents and stent thrombosis rate < 1%.
Study Design
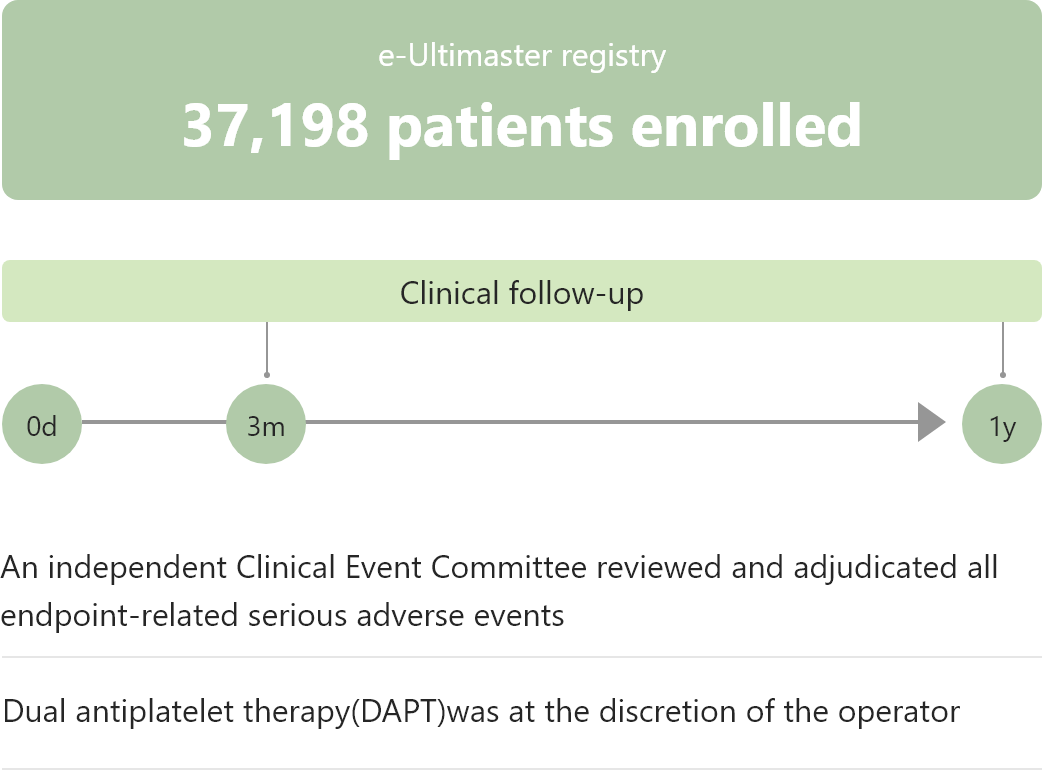
Primary Endpoint
Target lesion failure at 1 year
・Cardiac death, target vessel MI or clinically driven target lesion revascularization
Secondary Endpoint
Safety
・Cardiac death/myocardial infarction
・Stent thrombosis (according to ARC definitions; acute, subacute, and late)
・Major vascular and bleeding complications
Efficacy and patient-oriented endpoints
・Target lesion revascularization (TLR)
・Target vessel failure (TVF)
- Cardiac death, target vessel MI or clinically driven target vessel revascularization
・Patient-oriented composite endopoint (POCE)
- All-cause death, any MI or any coronary revascularization
Patient and lesion characteristics
Patient background
| Male | 76% |
|---|---|
| Diabetes | 28% |
| Smoking | 24% |
| Hypertension | 68% |
| Hypercholesterolemia | 60% |
| BMI≧30 | 25% |
| Age | 64 years |
| CCS | 45% |
| NSTE-ACS | 36% |
| STEMI | 20% |
| Radial access | ||
|---|---|---|
| CCS | 80% | |
| NSTE-ACS | 84% | |
| STEMI | 83% | |
| DAPT at 1 year | ||
| All patients | 67% | |
| CCS | 65% | |
| NSTE-ACS | 68% | |
| STEMI | 69% |
Result
5-year clinical outcomes